
Vol. 41, n.º 4, 2008
REVISTA
ESPAÑOLA DE
Vol. 41, n.º 4, 2008 |
CASUÍSTICA
Emilio Mayayo Artal
Servicio de Patología. Hospital Universitario
Juan XXIII y Departamento de Ciencias Médicas Básicas.
Facultad de Medicina. Universidad Rovira y Virgili. Tarragona.
emilio.mayayo@urv.cat
SUMMARY
Introduction: We present an unusual case of true synovial metaplasia of a periprosthetic breast capsule in a young woman with recidivant tumor. Material and methods: In a 31-year-old-female with breast tumor a surgical procedure was realized. Subcutaneous mastectomy and silicone prosthesis implant were performed due to a myxoid recidivant tumor, myxoid variant of malignant fibrous histiocytoma. Surgical excision twice revealed a complete sacular capsule surrounding the breast prosthesis adjacent to a recidivant tumor. Results: Histopathological study revealed that the membrane was covered by true metaplasia with two cell types. Immunohistochemical techniques were positive for CD-68, vimentin and S-100 protein. These results were correlated with ultrastructural demonstration of two different cell types. X-ray microanalysis demonstrated the presence of silicone at the surface and within the membrane, indicating that metaplasia was triggered by silicone and not by the surgical procedure, as described in other locations. Conclusions: On basis of the results presented, we consider that this lesion is a true synovial metaplasia to silicone prosthesis. There are not more than five cases with these characteristics in medical literature.
Keywords: Breast, synovial metaplasia, immunohistochemical study, silicone.
RESUMEN
Introducción: Presentamos un caso excepcional de metaplasia sinovial localizada en la cápsula producida como respuesta a prótesis mamaria de silicona como consecuencia de un raro tumor recidivante. Material y métodos: Mujer de 31 años a la que se realiza mastectomía e implante de prótesis de silicona, por tumor mixoide recidivante diagnosticado como histiocitoma fibroso maligno, variante mixoide. Por dos veces se implantó prótesis de silicona y ambas produjeron una cápsula integra fibrosa. Resultados: El estudio histopatológico evidenció que la membrana estaba revestida por una verdadera metaplasia sinovial con dos tipos celulares. Las técnicas de inmunohistoquímica fueron positivas para CD-68, vimentina y proteína S-100. Esto se correlacionó con la ultraestructura donde se ponen de manifiesto dos tipos bien diferenciados de células. Además se realizó microestudio de partículas y se pudo observar presencia de silicona en la superficie de la membrana. Conclusiones: Los resultados presentados nos manifiestan que la silicona es el material desencadenante de la metaplasia sinovial y no las maniobras quirúrgicas como están descritas en otras localizaciones. Se trata de metaplasia sinovial verdadera siendo menos de cinco los casos publicados en la literatura médica.
Palabras clave: Mama, metaplasia sinovial, inmunohistoquímica, silicona.
INTRODUCTION
Synovial metaplasia is an exceptional pathology. It was reported previously in lesions of the soft tissue around the joints on sialastic rod or prosthetic materials or produced experimentally following repetitive subcutaneous injections of air after surgical procedures (1-3). Three cases of synovial metaplasia of the skin in association with surgical procedure have been reported by Gonzalez et al (4). Presently only nine cases more have been described, some of them with the name cutaneous metaplastic synovial cyst (5-8).
The first case of synovial metaplasia in breast was reported in a 48-year old woman secondary to silicone breast prosthesis by Raso et al (9). Later, these authors made a complete revision of fifty periprosthetic breast capsule cases and reported an additional one of synovial metaplasia (10). Recently, some new cases have been reported (11-15). We present a new case of a complete and true synovial metaplasia of breast which an extensive histological, immunohistochemical and ultrastructural study was carried out.
CASE REPORT
A 31-year-old woman was surgical treated in five occasions (the first at 28 years) due to the development of a recidivant tumor in her left breast. The histopathological study revealed a mixoma. Subcutaneous mastectomy followed by reconstruction with 290 ml textured silicone prosthesis was performed. The six removed tumors were classified as myxoid fibrous histiocytoma with a low grade of malignancy, which is under study at present time. Nine months later, new lumpectomy was done due to a recidivant tumor, removing the capsule but not the prosthesis. Histopathological study revealed similar previous findings at the tumor as well as the presence of synovial metaplasia at the capsule. One year later, a new tumor with cutaneous affection was observed. Removal of the new recidivant tumor and of the affected skin tissues was carried out. The silicone prosthesis was extracted together with a free intracapsular nodule.
Pathological Findings
A silicone breast prosthesis had induced twice a clear response of the body, producing an integral envelope of sacular type with a brilliant surface in contact with the prosthesis and not at the side of the tumor (fig. 1). Microscopically, three histological layers were identified: intimae, subintima and fibrous capsule (fig. 2). The intimae showed a varying amount of fibrin, while the cellular sheaths (subintima) were constituted of different layers of mononuclear and multinuclear cells (fig. 3). The latter are normally found in the deepest layers and in contact with the neoformed capillars (fig. 4). The intracapsular free nodule is formed by a carpet-like structure with eroded areas and fibrin. Giant multinucleated cells with birrefrigent cytoplasmic inclusions are also observed. Similar cells are detected in the deep stroma of the capsule. Alcian-blue stained the intimae extensively while PAS was negative due to the absence of a basement membrane. Gomori’s reticulin stain revealed a prominent network of reticulin fibers surrounding the individual lining cells.
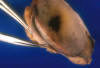
Fig. 1:
Open sacular capsule and detail of the tumor near to silicone breast prosthesis.
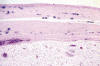
Fig. 2:
Surface cellular membrane separating mass with a well vascularised band (H-E
x80).
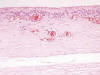
Fig. 3:
Synovial metaplasia of surface membrane (H-E x250).
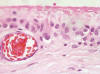
Fig. 4:
Detail showing two-cell type, with surface fibrin apposition (H-E x400).
An immunohistochemical study with a broad spectrum of antibodies as cytokeratin, EMA, vimentin, S-100 protein, CEA, lysozime, a-antichimotrysin, a-antitripsin, desmin, and CD-68, was performed on section that had been fixed in 10% neutral buffered formalin solution and embedded in paraffin. Vimentin, S-100 protein and CD-68 were strongly positive. Lysozime, a-antitripsin, and a-antrichimotripsin showed less intense staining while the other antibodies were negative. For transmission electron microscopy, preparates, fixed in formalin as described previously, were postfixed in 1% osmium tetroxide, dehydrated and embedded in Spur’s medium.
Observations were performed at 80 Kv in a Zeiss EM-10. For scanning electron microscopy, a similar procedure of fixation was performed on the samples followed by dehydration and critical point drying. After gold sputter coating, specimens were observed in a Jeol JSM6400. With the same instrument fitted with a Link analyser eXL II, X-ray microanalysis was performed on carbon coated preparates where osmium tetroxide was avoided.
The electron microscopy results revealed two well defined types of cells loosely arranged. Scanning electron microscopy demonstrated the formation of a synovial organized in a carpet-like structure, with a fibrous under laying tissue. Presence of inorganic deposits was detected at the surface of the synovial layer. The chemical composition of these deposits revealed presence of natrium, silicon phosphorus, sulphur, cloride and calcium. Similar spectra were observed within the synovial layer at the areas free of inorganic deposits.
DISCUSSION
The first two cases of synovial metaplasia of a periprosthetic silicone breast capsule have reported by Raso et al (9,10). In their broad revision of fifty breast prosthesis, they attributed the cause o induction of synovial metaplasia more to the silicone than to the surgical procedures and they pointed out the need for further investigation to clarify its origin and cause (10). More recently short series of synovial-lyke hyperplasia (12,13) or pseudoepithelization (11) to breast implant capsules have been published.
The deceptive name of synovial metaplasia is only based on the morphological a resemblance of villous structures of the hyperplasic synovial membrane at histological studies (4,16). However, a broad spectrum of possible histological appearances may appear; from incomplete to complete (5). Histologically, two types of cells have been observed: Type A and Type B, basically differentiated (4,8) by varying amounts of cytoplasmic vacuoles, pinocytotic vesicles and rough endoplasmic reticulum (8,17,18). Similar cells have been seen in silicone-rod-induced pseudotendon sheaths and in synovial membrane (8,17,18). In recent studies it has been suggested that the type-A cells are derived from bone marrow and the type-B cells from local mesenchymal cells (4,5,8). The immunohistochemical studies performed here, demonstrate that the type-B cells are basically positive for vimentin. This type of cell is the more abundant in incomplete forms of synovial metaplasia. The type-A cells are typical on the complete synovial metaplasia. They are positive to CD-68 and stain for macrophage-associated antigens, which are usually seen in synovium. They also react for S-100 protein and lysozyme (4,19,20). Recently, Hammed et al (13) also proved positivity to S-100 protein and vimentin for the synovial-lyke membranes as in our case. Furthermore, they found that concavalin A and peanut agglutinin, lectings that bind to cell-surface receptors of histiocytes, are also positive. These findings would corroborate the positivity obtained for CD-68 in our study. For the confirmation of these immunohistochemical findings, several controls with normal and hyperplastic synovials were carried out, which also showed to be positive to CD-68, S-100 protein, vimentin and lysozime (19,20). This positivity, together with the ultrastructural pattern, could confirm the hypothesis that the present case is a true synovial metaplasia to periprosthetic breast capsule.
At the ultrastructural study, two different types of cells, similar to the ones described by other authors were observed (9). The organization in a carpet-like structure of the synovial cells with an under laying fibroses capsule, formed by parallel arranged collagen fibbers, was comparable to the structure reported by Raso et al (9,10). A finding not recorded before, is the presence of inorganic deposits on the surface of the synovial layer. The nature of which showed the presence of silicone among other elements with SEM X-ray microanalysis. This technique in combination with the transmission electron microscope has been previously used to demonstrate the presence of silicone deposits within vacuoles of phagocytic cells lining in contact with the prosthesis as well as in the deep fibrous extracellular matrix (14). Apart from the silicone, we also detected other elements, such as phosphorous and sulphur. They were not described previously due to the inadequate preparation of the samples that included osmium in the fixation procedure (9,14). From our point of view the elements detected can be part of the prosthesis and be captures, we can conclude a true synovial origin for this unusual case of breast periprosthesis capsule. A point that remains unsolved is the reasons that produce this unusual breast metaplasia, since in this location this type of cells is absent. It is only possible to suggest that under certain stimulus, migration from the mesenchyme or from bone marrow of these cells occur. However, it can also be possible that certain transformations from stem cells could exist. Therefore, further studies will be needed to clarify this point.
REFERENCES
Edwards JC, Sedgwick AD, Willoughby DA. The formation of a structure with the features of synovial lining by subcutaneous injection of air: an in vivo tissue cultures system. J Pathol 1981; 134: 147-56.
Goldring SR, Schiller AL, Roelke N, Rourke CM, O’Neill DA, Harris WH. The synovial-like membrane at the bone-cement interface in loose total hip replacements and its proposed role in bone lysis. J Bone Joint Surg Am 1983; 65: 575-84.
Hunter JM, Jaeger SH, Matsui Y, Miyaji N. The pseudosynovial sheath: its characteristics in a primate model. J Hand Surg 1983; 8: 461-70.
Gonzalez JG, Ghiselli RW, Santa Cruz DJ. Synovial metaplasia of the skin. Am J Surg Pathol. 1987; 11: 343-50.
Beham A, Fletcher CD, Feichtinger J, Zelger B, Schmid C, Humer U. Synovial metaplasia of the skin. Virchows Archiv A Pathol Anat 1993; 423: 315-8.
Bhawan J, Dayal Y, Gonzalez-Serva A, Eisen R. Cutaneous metaplastic synovial cyst. J Cutan Pathol 1990; 17: 22-6.
Gomez-Dorronsoro ML, Martinez-Peñuela JM, Ruiz de la Hermosa J. Metaplastic synovial cyst. Am J Surg Pathol 1988; 12: 649-50.
Stern DR, Sexton RM. Metaplastic synovial cyst alter partial escision of nevus sebaceus. Am J Dermatopathol 1988; 10: 531-5.
Raso DS, Green WB, Metcalf JS, Synovial metaplasia of a periprosthetic breast capsule. Arch Pathol Lab Med 1994; 118: 249-51.
Raso DS, Crymes LW, Metcalf JS. Histological assessment of fifty breast capsules from smooth and textured augmentation and reconstruction mammoplasty protheses with emphasis on the role of synovial metaplasia. Mod Pathol 1994; 7: 310-6.
Emery JA, Spanier SS, Kasnic G Jr, Hardt NS. The synovial structure of breast-implant-associated bursae. Mod Pathol 1994; 7: 728-33.
Del Rosario AD, Bui HX, Petrocine S, Sheeham C, Pastore J, Singh J, Ross JS. True synovial metaplasia of breast implant capsules: a light and electron microscopy study. Ultrastruct Pathol 1995; 19: 83-93.
Hameed MR, Erlandson R, Rosen PP. Capsular synovial-like hyperplasia around mammary implants similar to detritic synovitis. A morphologic and immunohistochemical study of 15 cases. Am J Surg Pathol 1995; 19: 433-8.
Raso DJ, Greene WB. Silicone breast implants: pathology. Ultrastruc Pathol 1997; 21: 263-71.
Ramidial PK, Sing Y, Singh B. Metaplastic synovial cyst in male breast. Ann Diagn Pathol 2005; 9: 219-22.
Henderson B, Pettipher ER. The synovial living cell: biology and pathobiology. Semin Arthritis Rheum 1985; 15: 1-32.
Barland P, Novikoff AB, Hamerman D. Electron microscopy of the human synovial membrane. J Cell Biol 1962; 14: 207-20.
Jilani M, Ghardially FN. An ultrastructural study of age associated changes in the rabbit synovial membrane. J Anat 1986; 146: 201-15.
Apte SS, Athanasou NA. An immunohistological study of cartilage and synovium in primary synovial chondromatosis. J Pathol 1992; 166: 277-81.
Athanasou NA, Quinn J, Heryet A, Puddle B, Woods CG, NcGee JO. The immunohistology of synovial lining cells in normal and inflamated synovium. J Pathol 1988; 155: 133-42.